Blueberry paste on treating experimental toxic hepatitis and intestinal dysbiosis
A.P.Levitsky, Dr.Sc. /Biology/, S.B.Osipenko, Candidate of Science /Technic/, Yu.V.Tsiselsky, Candidate of Science /Medicine/, S.A.Demianenko, Candidate of Science /Mediсine/, O.A.Makarenko , Candidate of Science /Biology/, I.A.Selivanskaia, Candidate of Science
Stomatology Institute, Ukrainian Academy of Medical Sciences, TEKMASH Scientific Industrial Private Institute, Kherson, Regional Clinical Hospital, Odessa, QUALITET Private Enterprise, Yevpatoria
HEPATOPROTECTIVE CHARACTERISTICS OF BLUEBERRY PASTE FOR TREATING EXPERIMENTAL TOXIC HEPATITIS AND INTESTINAL DYSBACTERIOSIS
Blueberries (Vaccinium myrtillus) are widely used in food and in folk medicine [1, 2]. As they contain a lot of biologically active substances, especially polyphenols, among which anthocyanins and other bioflavonoids [3, 4] constitute the greatest part, blueberries are famous for their antioxidant, anti-inflammatory, hypoglycemic and antimicrobial effect [5]. Owing to it, blueberries are used as decoctions, tinctures, syrups, kissels in treating pancreatic diabetes, arthritis, podagra, colitis, eye diseases, etc. Quite often they are used together with other plants [6, 7]
Unfortunately, there has not been much research concerning the expediency of blueberry use in treating hepatobiliary pathology [8]. Thus, considering a great number of polyphenols /bioflavonoids, in particular/ in blueberries and their hepatoprotective effect [9] decision has been made to study blueberry paste medioprophylactic effect when modeling toxic hepatitis against intestinal disbacteriosis which tends to aggravate the liver condition [10].
MATERIALS AND METHODS
30 adult white female rats of Wistar line /aged 13 months, weighing 300 20 g/ have been used for the experiment. They have been divided into 3 groups, 10 rats in each. The first group being the control one, the second and the third groups reproduced the liver combined pathology: toxic hepatitis as a result of single intraperitoneal introduction of 50% carbon tetrachloride oil solution /1ml/ and intestinal dysbacteriosis as a result of introducing lincomycin with cooking soda /60 mg per 1 kg of live weight/ for 5 days [12].
For four days before introducing hepatitis and dysbacteriosis, group 3 had been daily fed 2 g of blueberry paste produced by original TEKMASH technology /Kherson/ [22]. All in all, the rats of this group had been supplied with blueberry paste for 12 days /4 days before modeling the pathology, 5 days during the modeling and 3 days after it until euthanasia/. Euthanasia was performed under thiopental anesthetic by the total bloodletting from the heart, the liver was removed and serum was obtained. Liver homogenate (50 mg/ml of 0.9% NaCl) was analyzed for malonic dialdehyde /MDA/ concentration [13], for general proteolytic activity /GPA/ [14], for alkaline phosphatase activity /AP/ [15] and catalase activity [16].
The blood serum was analyzed for MDA concentration, for total bilirubin [17], for GPA, for alaninetransaminase activity /ALT/ [18], for AP and catalase. After that antioxidant-prooxidant index API [19] was calculated by relating catalase activity to MDA concentration.
Blueberry paste chemical analysis was made using the following methods: mass fractions of moisture, protein, fiber, sugar, general carbohydrates, fructose, carotenoids were analyzed according to [20], polyphenol mass fractions - according to [21].
RESULTS AND DISCUSSION
The results of the chemical analysis of natural blueberries and blueberry paste made by TEKMASH Institute technology are given in Table 1. As the table indicates, the paste preserves all biochemical characteristics of natural berries, only protein and fiber concentrations being reduced. Blueberry paste polyphenolic content remains the same and equals 7900 mg/kg. One can hardly find another plant containing such a great amount of polyphenolic compounds, anthocyanins constituting the majority [4].
Table 1 Chemical composition of natural blueberries and blueberry paste
| Characteristics | Natural blueberries | Blueberry paste |
| Moisture, % | 84,1 | 8,4 |
| Protein, % | 0,67 | 0,35 |
| Fibre, % | 2,5 | 1,5 |
| General carbohydrates, % | 13,5 | 13,0 |
| Including sugar, % | 9,1 | 9,1 |
| Including fructose and fructosydes | 5,6 | 5,4 |
| Polyphenols, mg/kg of dry matter | 6900 | 7900 |
| Carotenoids, mg/kg of dry matter | 8,0 | 7,2 |
Note: Average of three determinations.
Tables 2 and 3 demonstrate the results of blood serum and liver tissue biochemical tests on experimental hepatitis and dysbacteriosis rats that were fed blueberry paste daily (2 g/day per rat). Liver characteristics are visualized in fig. 1. The control group characteristics are taken as 100%. All biochemical markers of liver pathology point to hepatic cells toxic involvement. However, feeding rats with blueberry paste contributes to depressed hepatotoxic characteristics both of liver tissue and blood serum.
Table 2 Liver biochemical characteristics of experimental hepatitis and dysbacteriosis rats fed blueberry paste (n=10, M m)
| Characteristics | Control (1) | Hepatitis+dysbacteriosis (2) | Hepatitis+dysbacteriosis against blueberry diet (3) |
| MDA, mmole/kg | 32,1 ± 4,1 | 53,4 ± 4,1 р < 0,001 |
42,4 ± 5,1 р > 0,1 р1 > 0,05 |
| GPA, /kg | 24,1 ± 4,0 | 39,1 ± 3,0 р < 0,05 |
26,5 ± 2,8 р > 0,4 р1 < 0,05 |
| AP, /kg | 3,76 ± 0,43 | 5,64 ± 0,68 р < 0,05 |
4,57 ± 0,42 р > 0,1 р1 > 0,05 |
| Catalase, /kg | 4,51 ± 0,10 | 4,01 ± 0,05 р < 0,01 |
4,34 ± 0,02 р > 0,05 р1 < 0,01 |
Notes:
p - confidence index compared to control group;
p - confidence index compared to group 2.
Table 3 Blood serum biochemical characteristics of experimental hepatitis and dysbacteriosis rats fed blueberry paste (n=10, M m)
| Characteristics | Control (1) | Hepatitis+dysbacteriosis (2) | Hepatitis+dysbacteriosis against blueberry diet (3) |
| MDA, mmole/kg | 0,77 ± 0,03 | 0,91 ± 0,04 р < 0,05 |
0,79 ± 0,06 р > 0,5 р1 > 0,05 |
| GPA, /kg | 2,40 ± 0,16 | 3,65 ± 0,44 р < 0,05 |
2,48 ± 0,32 р > 0,6 р1 < 0,05 |
| Total bilirubin, | 4,71± 0,71 | 8,49 ± 1,41 р < 0,05 |
6,18 ± 1,59 р > 0,3 р1 > 0,05 |
| ALT | 0,14 ± 0,04 | 0,24 ± 0,06 р > 0,1 |
0,12 ± 0,03 р > 0,5 р1 > 0,05 |
| AP | 1,48 ± 0,23 | 2,10 ± 0,21 р > 0,05 |
1,68 ± 0,19 р > 0,3 р1 > 0,05 |
| Catalase | 0,323 ± 0,004 | 0,303 ± 0,005 р < 0,01 |
0,314 ± 0,005 р > 0,1 р1 > 0,05 |
Notes:
1. p - confidence index compared to control group;
2. p1 - confidence index compared to group 2.
Table 3 Blood serum biochemical characteristics of experimental hepatitis and dysbacteriosis rats fed blueberry paste (n=10, M m)
| Characteristics | Control (1) | Hepatitis+dysbacteriosis (2) | Hepatitis+dysbacteriosis against blueberry diet (3) |
| MDA, mmole/kg | 0,77 ± 0,03 | 0,91 ± 0,04 р < 0,05 |
0,79 ± 0,06 р > 0,5 р1 > 0,05 |
| GPA, /kg | 2,40 ± 0,16 | 3,65 ± 0,44 р < 0,05 |
2,48 ± 0,32 р > 0,6 р1 < 0,05 |
| Total bilirubin, | 4,71± 0,71 | 8,49 ± 1,41 р < 0,05 |
6,18 ± 1,59 р > 0,3 р1 > 0,05 |
| ALT | 0,14 ± 0,04 | 0,24 ± 0,06 р > 0,1 |
0,12 ± 0,03 р > 0,5 р1 > 0,05 |
| AP | 1,48 ± 0,23 | 2,10 ± 0,21 р > 0,05 |
1,68 ± 0,19 р > 0,3 р1 > 0,05 |
| Catalase | 0,323 ± 0,004 | 0,303 ± 0,005 р < 0,01 |
0,314 ± 0,005 р > 0,1 р1 > 0,05 |
Notes:
1. p - confidence index compared to control group;
2. p1 - confidence index compared to group 2.
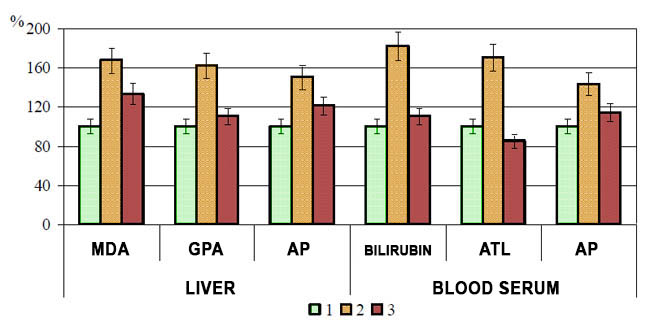
Fig. 1 Blueberry paste effect on liver biochemical characteristics of experimental hepatitis and dysbacteriosis rats
(1 - control, 2 - hepatitis+dysbacteriosis, 3 - hepatitis+dysbacteriosis against blueberry paste diet
Blueberry medioprophylactic effect can be attributed to its polyphenolic compounds influencing the body antioxidant systems estimated by API index /fig.2/. As illustrated in the figure, the blueberry diet reliably increases this index which is reduced by the liver pathology both in the liver and in the serum.
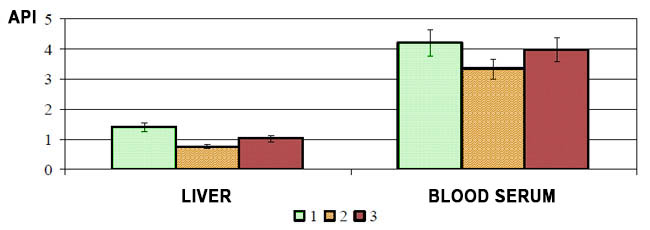
Fig. 2. Blueberry paste effect on API index in liver and blood serum of experimental hepatitis and dysbacteriosis rats (1 - control, 2 - hepatitis+dysbacteriosis, 3 - hepatitis+dysbacteriosis against blueberry paste diet)
Thus, our investigation has shown blueberry high hepatoprotective effect even with serious liver pathology caused by CCl intoxication and intestinal dysbacteriosis.
In our opinion, blueberry high medioprophylactic potential has been greatly underrated by the official medicine though it has been widely used by folk medicine.
CONCLUSIONS
1. Combined liver pathology (carbon tetrachloride intoxication and intestinal dysbacterios) causes serious hepatic cells involvement which is reliably defined by the model under study and biochemical markers.
2. Blueberry paste made by TEKMASH technology practically eliminates many symptoms of the liver pathology, which testifies to the expediency of studying its hepatoprotective characteristics for further clinical use.
REFERENCES
1. Dudchenko N.G., Krivenko V.V. Food plant healers //K.: Naukova dumka, 2nd publication, 1988.-272 p.
2. Formazuk V.I. Food medicinal plants encyclopaedia. Cultivated and wild plants in practical medicine // K.: A.C.K., 2003. - 792 p.
3. Kolesnikov M.P., Gins V.K. Phenolic compounds in medicinal plants // Applied biochemistry and microbiology. - 2001. - V. 37, # 4. - P.457-465.
4. Andersen O.M., Markhan K.R. Flavonoids: Chemistry, Biochemistry and Applications // Taylor and Francis CRC Press., 2005. - 1256 p.
5. Petkov V. (edit.) Modern phytotherapy. Sofia // Medicine and physical culture. - 1988. - p. 302-303.
6. Demchenko D.V., Panok Yu.D., Legosteva A.B. Development of liquid extract technology using blueberry and ginseng leaves as the base // Chemical pharmaceutical journal. - 2008. - V. 42, # 3. - P. 20-24.
7. Nikitina L.K. General health improving and tonic remedy and its production method // Patent 2321418 Russia MPC A61K 36/00 A61P 43//00. # 2006138357/15. - Applic. 30.10.2006; Publ. 10.04.2008.
8. Karhut V.V. Live pharmacy // K.: zdorovia, 1992. - 312 p.
9. Dorkina Ye.G. Natural flavonoid compounds hepatoprotective action study // Experimental and clinical pharmacology. - 2004. - V. 67, # 6. - P. 41-44.
10. Wigg A.J., Robert-Thompson J.G., Dymock R.B. The role of small intestinal bacterial overgrowth, intestinal permeability, endotoxaemia and tumor necrosis factor-alfa in a pathogenesis of nonaleocholic steatohepatitis // Gut. - 2001. - V. 48. - P. 206-211.
11. Stefanov O.V. (edit.) Pre-clinical remedy study // K.: DFC. - 2001. - P. 334-351.
12. Levitsky A.P., Selivanska I.O., Tsiselsky Yu.V., Pochtar V.M., Rozsahanova L.M., Gulavsky V.T. Dysbacteriosis modeling method // Design patent # 31012, UA MPC (2006) A61P 31/00. - 2008. - Bulletin # 6.
13. Stalnaia I.D., Garishvily T.G. Method of malonic dialdehyde determination with the help of thiobarbituric acid // In book: "Modern biochemical methods: M. Medicine, 1977. - P. 66-68.
14. Levitsky A.P., Konobets V.M., Lvov I.F., Barabash R.D., Volodkina V.V. Kallikreins and nonspecific proteases in the saliva of gastric and duodenal ulcer patients // Medical chemistry problems. - 1973. - V. 19, # 6. - P. 633-638.
15. Levitsky A.P., Marchenko A.I., Ribak T.L. Comparative characteristics of three methods for defining human saliva phosphatases // Labor. Business. - 1973. - # 10. - P. 624-625.
16. Karoliuk M.A., Ivanova L.I., Maiorova N.T., Tokarev K.Ye. Method of defining catalase activity. - Labor. Business. - 1988. - # 1. - P. 16-18.
17. Goriachkovsky A.M. Clinical biochemistry in laboratory diagnostics. 3rd publ. // Odessa: Ecology, 3rd publ., 2005. - 616 p.
18. Reitman S., Frankel S.A. A colorimetric method for the determination of serum glutamic oxalaceric and glutamic purivic transaminases // Amer. j/ Clin. Pathol. - 1957. - V. 28, # 1. - P. 56-63.
19. Levitsky A.P., Pochtar V.M., Makarenko O.A., Gridina L.I. Blood serum antioxidant-prooxidant index of experimental stomatitis rats and its correction by dentifrice waters // Odessa medical journal. - 2006. - # 1. - P. 22-25.
20. Yermakov A.I. (edit.) Plants biochemical research methods // Leningrad: VO Agropromizdat, 3rd publ., 1987. - 430 p.
21. Martinchik E.A., Baturin A.K., Kosheleva O.V., Tutelian V.A. Flavonoids determination in fruit and vegetables and principles of creating design database for estimating flavonoids consumption by people // Nutrition problems. - 2006. - V. 75, # 6. - P. 34-37.
22. Osipenko S.B. "Method of seeds dispergation and equipment for it". Eurasian patent # 007199. Priority of 23.12.2002 in Ukraine 3 200500843.
23. www.tekmash.ua
HEPATOPROTECTIVE CHARACTERISTICS OF BLUEBERRY PASTE FOR TREATING EXPERIMENTAL TOXIC HEPATITIS AND INTESTINAL DYSBACTERIOSIS
A .P. Levitsky, S.B.Osipenko, Yu.V.Tsiselsky, S.A. Demianenko, O.A.Makarenko, I.A. Selivanskaia
Blueberry paste made by TEKMASH technology has been studied for its effect on the liver condition in rats with modeled toxic hepatitis /CCl introduction/ and intestinal dysbacteriosis /lincomycin introduction/. It has been found that feeding blueberry paste /2 g per a 300 g rat/ is conducive to lower liver and serum malonic dialdehyde concentration, lower protease and alkaline phosphatase activity, lower serum bilirubin concentration and ALT. All this bears witness to blueberries and blueberry paste hepatoprotective effect.
- TEK-PD cavitation-type dispersing pump
SIPE TEKMASH Institute offers dispersing pumps for making fine-dispersed food emulsions and suspensions, 10-25 m³/h capacity.
More... - Feedstock feeding equipment. Milk substitute.
The site contains information on fattening animals, on soy paste properties and soybean processing technology (animal feeding).
More... - Condensed milk line TEK-CML
Condensed milk production line
More...
- IndiaSASIL AGRO LLP
'Kshiti', 127/2, Rockel Lane,
Vakhar Bhag,
Sangli - 416416
MAHARASHTRA, India
Shreyans Shah
tel. +91-9011068877
e-mail: sasilexim@gmail.com - Colombia & LA
KAVITEC S.A.S.

Carrera 16 # 9-68
Dosquebradas, Risaralda, Colombia.?ngela Mar?a Sanz E
Gerente Comercial
email:
angela.sanz@kavitec.co
Tel?fono: 3306102
Cel: 3043289012 - Ukraine